Electroorganic synthesis is a promising technique for realizing sustainable transformations and discovering new reactivities, but the added complexity of electrochemical systems can complicate the screening of reaction conditions. To address those issues, we have developed an automated microfluidic platform allowing high-throughput experimentation (HTE) electroorganic synthesis in 15 microliter droplets. This multifunctional platform enables rapid and material-efficient electrochemical reaction condition screening and measurement of reaction kinetics. Interdigitated electrodes allow high current density and efficient inter-electrode molecular transport,
facilitating exploring optimal operation conditions for electrochemical radical–radical cross-coupling reactions. In addition, microliter scale cyclic voltammetry enables rapid measurements of kinetics.
We have also developed a parallel plate microfluidic redox-neutral electrochemistry platform with broad applications in single-electron transfer chemistry, including radical-radical crosscoupling, Minisci-type reactions, and nickel-catalyzed C(sp2)–O cross-coupling. The cathode and anode simultaneously generate the corresponding reactive intermediates, and selective coupling of radicals is enabled by the rapid molecular diffusion across a microfluidic channel, outpacing the decomposition of the radical intermediates. Moreover, we have devised a three-layer stacked microfluidic flow cell to increase productivity and demonstrate the scalability of the method.
We have recently used our microfluidic systems in machine-learning-guided workflows to discover new electrochemical reactions. Experimental data were generated with the automated droplet platform and used to train machine learning models to categorize competent reactions. Parallel plate reactors operated with recirculating fluids allowed the synthesis of enough materials to characterize the discovered reactions. We are currently exploring additional electrochemical reactions, including reactions involving solids that droplet or parallel plate systems cannot handle.
Selected publications:
- Mo, G. Rughoobur, A.M K. Nambiar, K. Zhang, and K.F. Jensen, A multifunctional microfluidic platform for high-throughput experimentation of electroorganic chemistry, Angew. Chemie Int. Ed., 2020, 59 (47) 20890-20894
- Mo, Z. Lu, G. Rughoobur, P. Patil, N. Gershenfeld, A. I. Akinwande, S.L. Buchwald, and K.F. Jensen, Microfluidic electrochemistry for single-electron transfer redox-neutral reactions, Science 2020, 368, 1352–1357
- F. Zahrt, Y. Mo, K.Y. Nandiwale, R. Shprints, E. Heid, K.F. Jensen, Machine Learning Guided Discovery of Electrochemical Reactions, J. Am. Chem. Soc. 2022, 144, 49, 22599–22610.
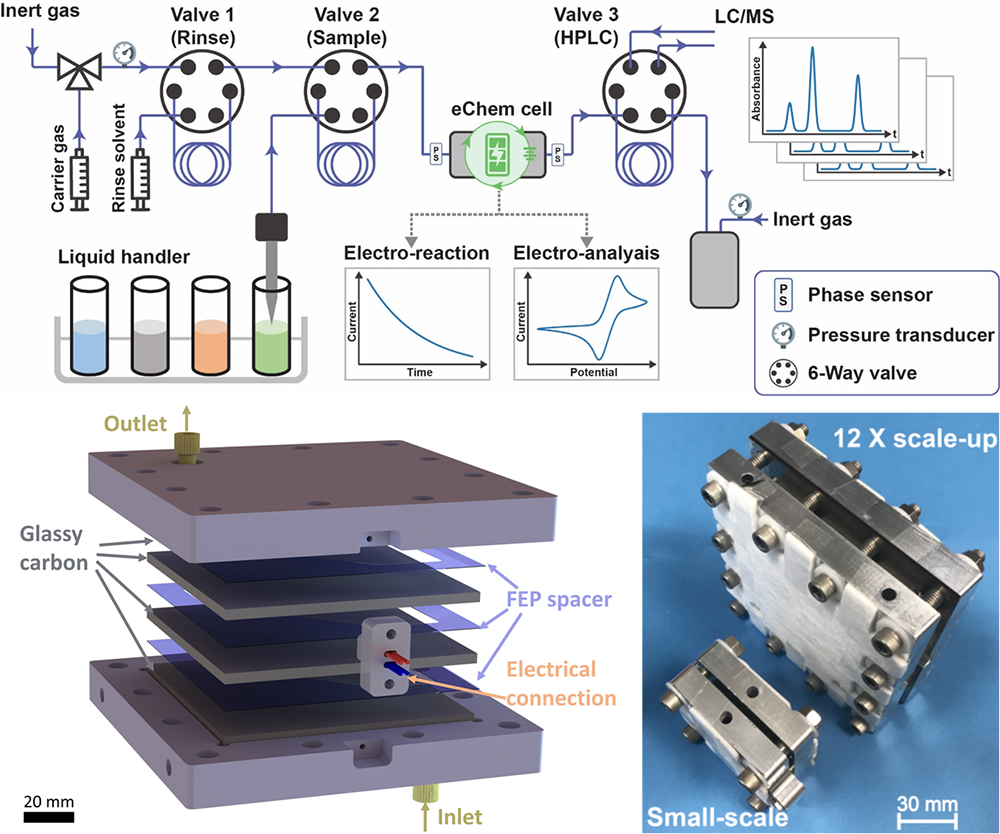
Top: The automated microfluidic platform for analysis, kinetics, and optimization of electroorganic reactions (Y. Mo et al. Angew. Chemie Int. Ed., 2020, 59 (47) 20890) Bottom: Parallel plate electrochemical reactors for redox-neutral electrochemistry. Left: schematic. Right: Images of actual reactors. (Y. Mo, et al. Science 2020, 368, 1352–1357)

